FDA puts Acomplia on slow track
As reviewed here at Junkfood Science, the full story on Acomplia, the new “miracle weight loss pill” being promoted by Sanofi-Aventis, is considerably different from that presented in the media. Perhaps the FDA reads this blog. :)
In a surprising move, the FDA has just put Acomplia on a slow track and deferred decision on whether to approve it until concerns over its side effects are reviewed by an FDA advisory panel on
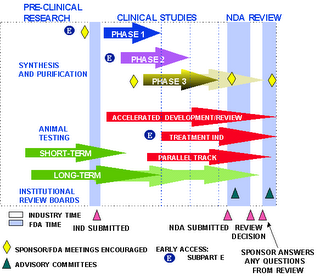
This is newsworthy because it runs contrary to the FDA’s new accelerated drug approval process for obesity and diabetes drugs initiated in 2003 under FDA commissioner Mark McClellan and Deputy Commissioner Lester Crawford. It was just one of the government’s comprehensive obesity initiatives. Under the FDA’s Obesity Working Group, created by McClellan and chaired by Crawford, a range of government programs were developed to educate the public of the dangers of obesity and role of diet and exercise, prioritize research and weight loss interventions, and encourage healthy foods.
The accelerated approval track under FDA guidelines has traditionally been used to speed to market drugs that treat life-threatening diseases on behalf of patients with low-survival expectations and no other treatment options. Over the past ten years, it has been used for drugs that treat things like AIDS and late-stage cancers. The FDA’s fast track can cut a year or more from the clinical trial phase of drug development and run these drugs through the FDA review period in as little as four months.
In a speech to PhRMA on March 28, 2003 McClellan told the pharmaceutical industry of these new regulatory and inspection efforts for obesity and diabetes drugs to “reduce the time and cost of product development.” As the Boston Globe reported, under this new approval process, it would also “allow drug companies to win licenses for new therapies based on preliminary indicators — measures such as weight loss or lower blood sugar — instead of waiting for completion of lengthy phase III clinical trials that show long-term effectiveness” in actual clinical outcomes.
Most consumers probably missed the story just two months ago when Lester Crawford, former FDA commissioner, pled guilty to a conflict of interest charge and for making false financial disclosures to the U.S. Senate and the Executive Branch. He’d failed to disclose significant financial interests in companies which stood to benefit from the recommendations of the FDA’s Obesity Working Group he oversaw. Crawford is scheduled to be sentenced on




<< Home